The PAIA glycan assay is the first product that enables researchers to tightly control changes of product glycosylation from initial clone selection through to large scale bioreactors – PAIA glycan assays are capable of analysing more samples in less time.
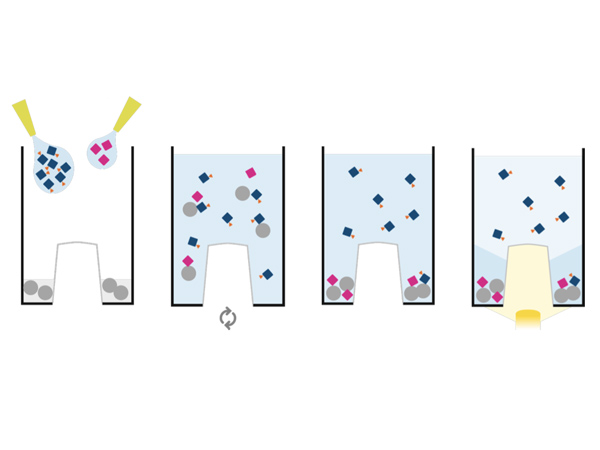
PAIA glycan screening assays (PA-201) detect the glycans on antibodies and Fc fusion proteins using capture beads that bind the analyte and labeled lectins that bind to specific types of glycans on the antibody.
PA-201 assay kits are customisable in that you select the type(s) of glycosylation you want to screen for and PAIA provide the assay kits containing the corresponding lectins.
PAIA glycan assays are easy to perform, demand significantly less sample preparation and material. Our customers save valuable time, usually several weeks, and gain the ability to asses a lot more samples than previously possible.
More on PAIA
PAIA Biotech has developed a high throughput platform technology for the quantification and glycan analysis of antibodies and proteins. Their proprietary based technology features bead-based immunoassays with fluorescence detection in specially designed 384-well microplates which drastically simplify the workflow. PAIA Biotech focuses on products for screening applications with limited sample volume and high throughput requirements, for example, IgG quantification. PAIA Biotech GmbH also offers custom assay development. PAIA Biotech GmbH was founded in 2014 and is based in Cologne.
Ellen’s take on this product
User friendly: Using the same technology as the antibody quantification assays, the plate-based glycan screening assays provide a fast and easy solution to analysing glycans during bioprocessing. This allows the researcher to monitor product quality earlier in the bioprocessing workflow.
Tailored:These easy-to-use kits come with everything you need to perform rapid screening for critical glycan features.
Did you know?
The PAIA assays can be quickly and easily measured using SYNENTEC’s NyONE and Cellavista imagers.
Make an enquiry
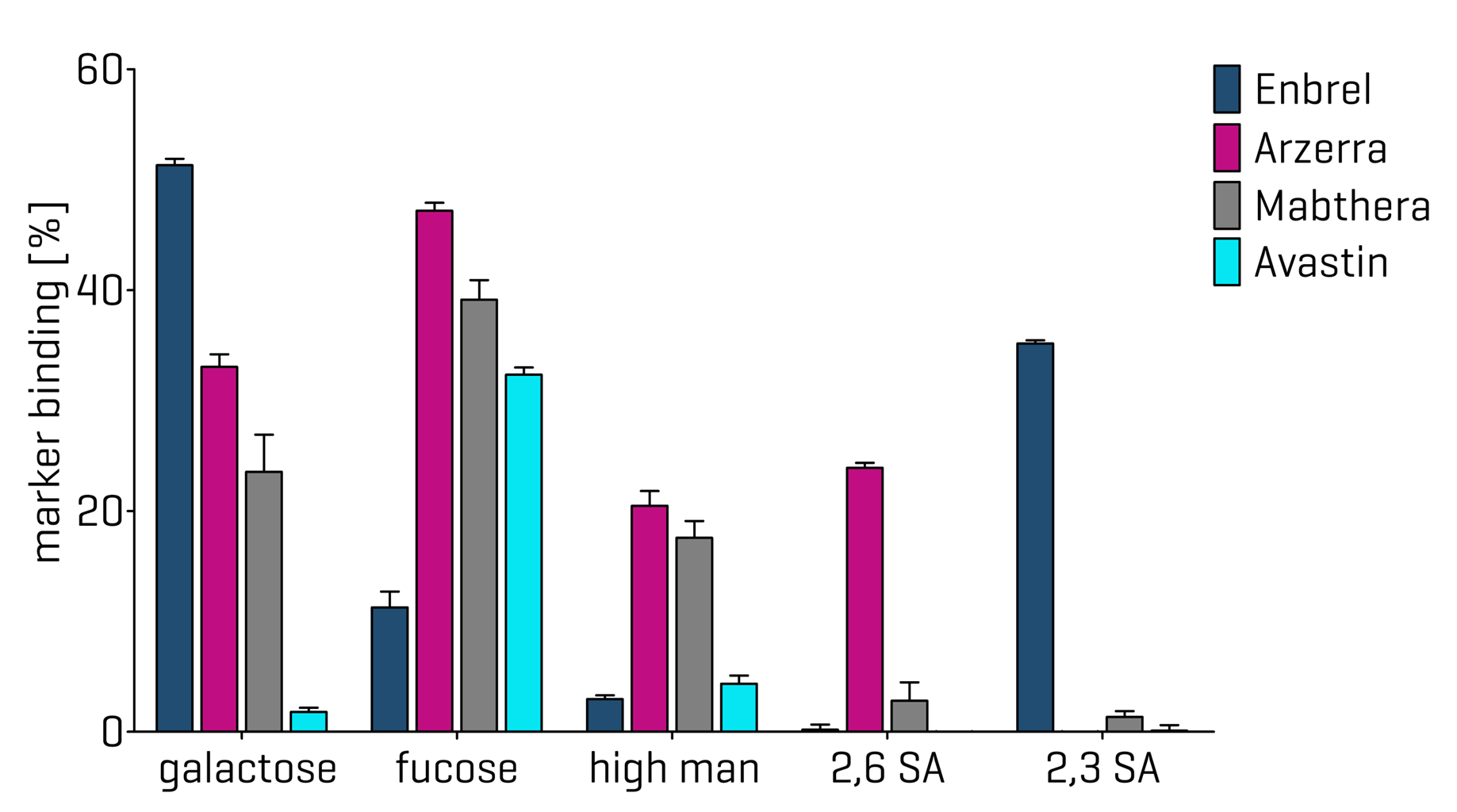
Lectins with defined specificities are available for core fucose, terminal galactose, high mannose (Man5/6) on N-linked glycans and for sialic acids on either N- or O-linked glycans.
The output of the PAIA glycan screening assay is a lectin binding rate for each of the lectins used. It represents the amount of lectin that has bound to the glycans on the antibody. Lectin binding profiles can be generated if several lectins are used in the assay to get an overview of the analyte´s overall glycosylation.
RESOURCES:
Antibody glycan screening
The most relevant glycosylation screenings on antibodies and bispecifics target Man5, core fucose and galactosylation, which are all located on the Fc domain of the molecule. Antibodies containing higher levels of Man5 are known to be eliminated from the blood faster and therefore considered as low-quality products. Furthermore, higher Man5 levels hint at unstable cell culture conditions.
Identification of low-quality clones with elevated Man5 levels in early development
Afucosylation is important if the product needs to evoke antibody dependent cell mediated cytotoxicity (ADCC). Antibodies lacking core fucose show an increased interaction with the FcγIIIa receptor and higher ADCC activity.
Assessment of afucosylation levels in products from genetically engineered cell lines
Galactosylation of an antibody is related to complement-mediated cytotoxicity (CDC). High levels of galactosylation cause higher CDC responses.
Selection of clones and media resulting in higher product galactosylation.



